Sperm are masters of tetris packing
Study sheds light on the process that plays a central role
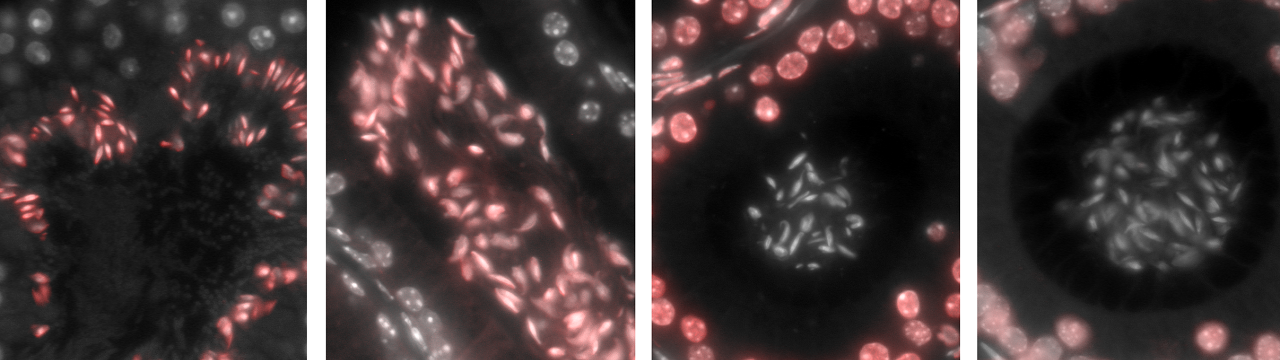
During sperm production, an enormous amount of DNA has to be packed into a very small space without breaking anything. A central role is played by certain proteins around which the DNA thread is wrapped – the protamines. A recent study by the University of Bonn provides new insights into this important mechanism. The results have been published in the journal PLoS Genetics.
If you are moaning once again about your suitcase being far too small as your vacation approaches, you should take human sperm cells as an inspiration. During their production, they are faced with an almost insoluble task. They have to pack 23 DNA threads with a total length of one meter into a head with a diameter of just three thousandths of a millimeter. And in the process, the delicate threads must not become entangled in an inextricable knot, nor must they tear.
We often sit on the suitcase to close it. The body resorts to a similar trick during spermatogenesis. Normally, DNA forms a comparatively loose tangle. In sperm cells, however, it is enormously compressed. ” If DNA were to take up as much space as a watermelon under normal circumstances, sperm cells would then only be as big as a tennis ball,” says Prof. Dr. Hubert Schorle from the Institute of Pathology at the University Hospital Bonn.
Biologists call this process hypercondensation. In their loose state, DNA threads are wrapped around numerous spherical protein molecules, the histones. In this state, they resemble 23 tiny strings of pearls. During hypercondensation, the histones are first exchanged for transition proteins. In a subsequent step, these are replaced by so-called protamines. Due to their chemical composition, protamines exert a very strong attraction on DNA. The thread therefore wraps itself in very firm and tightly loops around the protamine. […]
Participating Core Facilities: The authors acknowledge the support from the Mass Spectrometry, Next Generation Sequencing, and Microscopy Core Facilities. Furthermore, the Gene Editing Core Facility is affiliated with this study.
Participating institutions and funding:
The study was supported by the German Research Foundation (DFG) and the FEMHABIL program of the University Hospital Bonn.
Publication: L. Arévalo et al.: Loss of the cleaved-protamine 2 domain leads to incomplete histone-to-protamine exchange and infertility in mice; PLOS Genetics; DOI: https://doi.org/10.1371/journal.pgen.1010272