Elevator helps bacteria to build an invisibility cloak
Study: Bacterial membrane transporter helps pathogens to hide from immune system
The transport of substances across the membrane into the cell is linked to specific membrane transport proteins. Researchers at the University Hospital Bonn (UKB) and the University of Bonn, in collaboration with an international team, have now succeeded in elucidating the molecular structure of a completely new class of such membrane transporters. In addition to the Bonn scientists, researchers from the University of York were also involved. The study has now been published in the journal Nature Communications.
Like all cells, bacteria are surrounded by a cell membrane. This thin layer of fat holds together the cell’s nutrients, genetic material and proteins, thus enabling it to survive. On the other hand, nutrients or molecules that serve as building blocks for the cell, for example, must be able to cross the membrane, otherwise the bacterium would literally starve to death. For this purpose, cells make use of so-called membrane transporters. These are protein molecules that are located in the cell membrane and use complicated movements to transport substances across the membrane barrier.
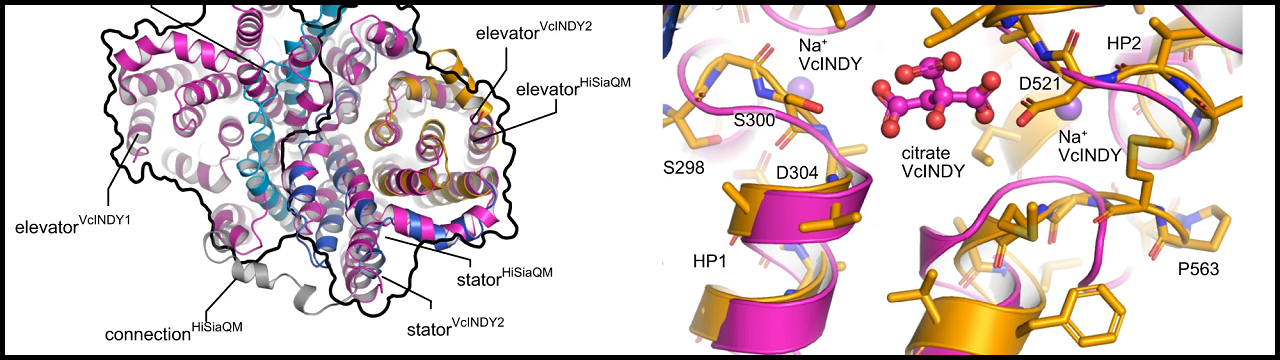
The so-called TRAP transporters help pathogens such as Haemophilus influenzae or Vibrio cholerae to move a small sugar molecule called sialic acid across the cell membrane. Sialic acid is very abundant in human tissue, and the bacteria can literally pick it up with the TRAP transporter and incorporate it into their cell wall. “There, it serves as a kind of invisibility cloak for the bacteria to hide from our immune system,” says Bonn scientist PD Dr Gregor Hagelueken.
The researchers from Hagelueken’s group at the Institute of Structural Biology at UKB used so-called cryo-electron microscopy to decipher the structure of the transporter. “Our results represent a real breakthrough, since attempts to decipher the structure of a TRAP transporter had already been going on for about 20 years,” says post-doctoral researcher Martin Peter from Bonn. […]
Participating Core Facilities: The authors acknowledge the support from the Nanobodies Core Facility.
Participating institutions and funding:
The Institute of Structural Biology and the Institute for Physical and Theoretical Chemistry were involved in the study. Partners were the Aston Centre for Membrane Proteins and Lipids Research (Birmingham, UK), the Department of Biology (Area 10) at the University of York (York, UK), the Biosciences Institute at the Newcastle University (Newcastle, UK), the Institut de Génétique et de Biologie Molecule et Cellulaire (Illkirch Cedex, France), and the Institute of Biophysics and Biophysical Chemistry at the University of Regensburg (Regensburg, Germany).
Publication: M. F. Peter et al.: Structural and mechanistic analysis of a tripartite ATP-independent periplasmic TRAP transporter; Nature Communications; DOI: https://doi.org/10.1038/s41467-022-31907-y